Bohrs model of the atom3/14/2024  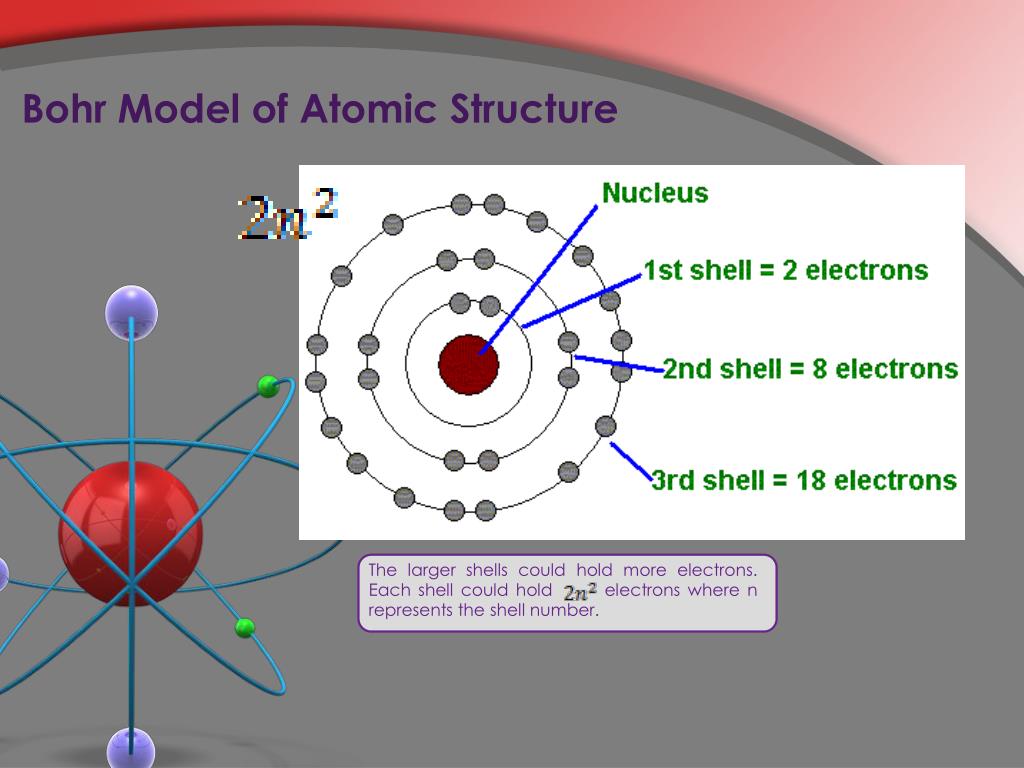
The vast majority of its mass is located in a positively charged central nucleus, which is 10,000. The Bohr model or Rutherford-Bohr model of the atom is a cake or planetary model that describes the structure of atoms mainly in terms of quantum theory. An electron can transition between orbits by absorbing or releasing energy that is exactly equal to the difference in energy of orbits, consistent with the law of conservation of energy. Heisenbergs uncertainty introduces to us a different concept entirely. Bohr’s model of the atom describes electrons orbiting in stable energy levels as opposed to Rutherfords model in which electrons motion was not described. It has a nucleus very precisely in the centre, and electrons in nice, neat orbitals around the outside, perfect circles with electrons moving around like planets. Rutherford had made the startling discovery that most of the atom is empty space. Bohrs model of the atom is all built out of things acting like golf balls. It comes to rest in the \(n = 6\) orbit, so \(n_2 = 6\). Bohr, one of the pioneers of quantum theory, had taken the atomic model presented a few years earlier by physicist Ernest Rutherford and given it a quantum twist. 
When atoms absorb energy, the electrons at a particular level are pushed up to higher levels (at bigger. In this case, the electron starts out with \(n = 4\), so \(n_1 = 4\). Bohrs solar system model of the atom is the way that most people think about atoms today. What is the energy (in joules) and the wavelength (in meters) of the line in the spectrum of hydrogen that represents the movement of an electron from Bohr orbit with n = 4 to the orbit with n = 6? In what part of the electromagnetic spectrum do we find this radiation? 
\): Calculating Electron Transitions in a One–electron System 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
